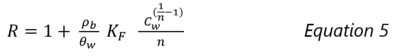Difference between revisions of "Sorption of Organic Contaminants"
Debra Tabron (talk | contribs) |
m (→Measuring Sorption) |
||
| (5 intermediate revisions by the same user not shown) | |||
| Line 4: | Line 4: | ||
'''Related Article(s):''' | '''Related Article(s):''' | ||
| + | |||
*[[Chlorinated Solvents]] | *[[Chlorinated Solvents]] | ||
*[[Petroleum Hydrocarbons]] | *[[Petroleum Hydrocarbons]] | ||
| Line 10: | Line 11: | ||
'''Key Resource(s)''': | '''Key Resource(s)''': | ||
| − | *[ | + | |
| + | *[//www.enviro.wiki/images/e/e8/1990-Piwoni-Basic_concepts_sorption_haz_site_EPA_540-4-90-053.pdf Basic Concepts of Contaminant Sorption at Hazardous Waste Sites]<ref>Piwoni, M.D. and Keeley, J.W., 1990. Basic concepts of contaminant sorption at hazardous waste sites. Ground-Water Issue; EPA-540/4-90/053. [//www.enviro.wiki/images/e/e8/1990-Piwoni-Basic_concepts_sorption_haz_site_EPA_540-4-90-053.pdf Report.pdf]</ref> | ||
==Introduction== | ==Introduction== | ||
| − | Sorption is a non-mechanistic term that describes uptake of a contaminant by a solid phase sorbent (soil, sediment, rock or an engineered material such as [ | + | Sorption is a non-mechanistic term that describes uptake of a contaminant by a solid phase sorbent (soil, sediment, rock or an engineered material such as [[wikipedia: Activated carbon | activated carbon]]). This article focuses on sorption of nonionic and nonpolar organic contaminants and their associations with subsurface materials. For these contaminants, sorption is almost exclusively associated with organic matter in the solids and is a reversible process. Sorption of ionic and polar organic contaminants is not described in this article. |
| − | In this article, the term ‘sorption’ includes all processes that cause the dissolved organic compound (sorbate) to become associated with the solid phase (sorbent). For nonpolar organic compounds, sorption occurs by either adsorption or absorption. [ | + | [[File:Allen-King1w2 Fig1a.png|right|thumb| 400 px|Figure 1a. Batch reactor experiments to generate points on a sorption isotherm (Figure 1b). This Figure shows two samples prepared in duplicate with soil-free control vials and vials to verify the contaminant mass added to the systems<ref name="Chiou1983">Chiou, C.T., Porter, P.E. and Schmedding, D.W., 1983. Partition equilibriums of nonionic organic compounds between soil organic matter and water. Environmental Science & Technology, 17(4), pp.227-231. [http://dx.doi.org/10.1021/es00110a009 doi: 10.1021/es00110a009]</ref>.]] |
| + | In this article, the term ‘sorption’ includes all processes that cause the dissolved organic compound (sorbate) to become associated with the solid phase (sorbent). For nonpolar organic compounds, sorption occurs by either adsorption or absorption. [[wikipedia: Absorption (chemistry) | Absorption]] is a volume-based process whereby the sorbate diffuses into the organic matter, analogous to the way that organic contaminants partition between immiscible oil and aqueous phases. In contrast, [[wikipedia: Adsorption | adsorption]] describes the interaction between the contaminant and the sorbent surfaces. | ||
==Measuring Sorption== | ==Measuring Sorption== | ||
| − | + | [[File:Allen-King1w2 Fig1b.png|thumb|400 px|Figure 1b. Linear isotherms for eight compounds in one soil<ref name="Chiou1983" />. ]] | |
| − | Sorption can be measured in the laboratory by placing a known mass of the contaminant, water, and uncontaminated sediment in a batch reactor ( | + | Sorption can be measured in the laboratory by placing a known mass of the contaminant, water, and uncontaminated sediment in a batch reactor (Figure 1a). The system is mixed for a specific duration (contact time) and then the aqueous and sorbed contaminant concentrations are determined. This process is repeated, adding a range of different initial contaminant masses to achieve a range of aqueous contaminant concentrations. When the experiment is run at constant temperature until the concentrations in the solid and liquid phases approach equilibrium, the resulting data plot is termed an isotherm. Figure 1b shows linear sorption isotherms for eight compounds measured in one soil. |
The time required to reach equilibrium can vary depending on the contaminant and sorbent. In studies with aquifer material from Canadian Forces Base Borden, tetrachloroethene (PCE) sorption reached equilibrium within 2 - 4 weeks, but tetrachlorobenzene did not reach equilibrium after 100 days. Powdered (pulverized) samples equilibrated more quickly indicating mass transfer within the grains was limiting<ref>Ball, W.P. and Roberts, P.V., 1991. Long-term sorption of halogenated organic chemicals by aquifer material. 2. Intraparticle diffusion. Environmental Science & Technology, 25(7), pp.1237-1249. [http://dx.doi.org/10.1021/es00019a003 doi:10.1021/es00019a003]</ref>. | The time required to reach equilibrium can vary depending on the contaminant and sorbent. In studies with aquifer material from Canadian Forces Base Borden, tetrachloroethene (PCE) sorption reached equilibrium within 2 - 4 weeks, but tetrachlorobenzene did not reach equilibrium after 100 days. Powdered (pulverized) samples equilibrated more quickly indicating mass transfer within the grains was limiting<ref>Ball, W.P. and Roberts, P.V., 1991. Long-term sorption of halogenated organic chemicals by aquifer material. 2. Intraparticle diffusion. Environmental Science & Technology, 25(7), pp.1237-1249. [http://dx.doi.org/10.1021/es00019a003 doi:10.1021/es00019a003]</ref>. | ||
| − | In some cases, the experimental data show an approximately linear relationship between aqueous and sorbed concentration, and the experimental results can be summarized in the form of a partition or distribution coefficient, ''K<sub>d</sub>'': < | + | In some cases, the experimental data show an approximately linear relationship between aqueous and sorbed concentration, and the experimental results can be summarized in the form of a partition or distribution coefficient, ''K<sub>d</sub>'': <br> |
| − | [[File:Allen-King1w2 Eq1.png|center| | + | [[File:Allen-King1w2 Eq1.png|center|320 px]] |
where ''C<sub>S</sub>'' is the solid (sorbed) concentration and ''C<sub>w<sub>'' is the aqueous (dissolved) concentration. By convention, ''C<sub>s</sub>'' has units of µg/g and ''C<sub>s</sub>'' has units of µg/mL, so ''K<sub>d</sub>'' has units of mL/g. | where ''C<sub>S</sub>'' is the solid (sorbed) concentration and ''C<sub>w<sub>'' is the aqueous (dissolved) concentration. By convention, ''C<sub>s</sub>'' has units of µg/g and ''C<sub>s</sub>'' has units of µg/mL, so ''K<sub>d</sub>'' has units of mL/g. | ||
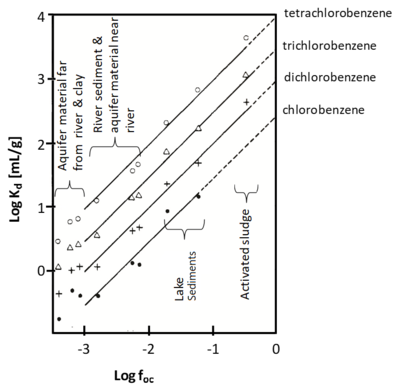
| − | When the relationship between ''C<sub>s</sub>'' and ''C<sub>w</sub>'' is non-linear, the experimental data are commonly represented by the Freundlich isotherm equation:< | + | When the relationship between ''C<sub>s</sub>'' and ''C<sub>w</sub>'' is non-linear, the experimental data are commonly represented by the Freundlich isotherm equation:<br> |
| − | [[File:Allen-King1w2 Eq2.png|center| | + | [[File:Allen-King1w2 Eq2.png|center|340px]] |
| − | where | + | where ''K<sub>F</sub>'' and ''n'' are empirically estimated coefficients specific to the sorbate, sorbent and equation used. |
| − | In most cases, ''absorption'' processes follow a linear isotherm. However, ''adsorption'' processes often follow a nonlinear relationship between the sorbed and dissolved contaminant concentrations. When both absorption and adsorption occur, a non-linear relationship is used to represent the sorption results. | + | In most cases, ''absorption'' processes follow a linear isotherm. However, ''adsorption'' processes often follow a nonlinear relationship between the sorbed and dissolved contaminant concentrations. When both absorption and adsorption occur, a non-linear relationship is used to represent the sorption results. |
==Impact on Groundwater Transport== | ==Impact on Groundwater Transport== | ||
Sorption reduces the amount of contaminant in the aqueous phase, slowing contaminant migration. Sorbed contaminants are also less available for biological or chemical degradation reactions, which will reduce the effectiveness of many remedial technologies. | Sorption reduces the amount of contaminant in the aqueous phase, slowing contaminant migration. Sorbed contaminants are also less available for biological or chemical degradation reactions, which will reduce the effectiveness of many remedial technologies. | ||
| + | |||
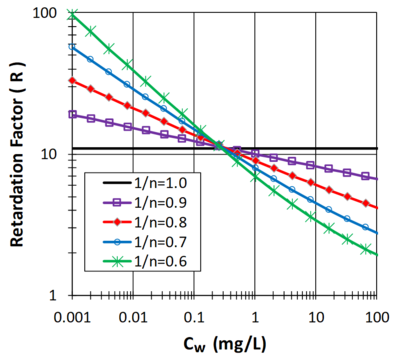
| + | [[File:Allen-King1w2 Fig2.png|thumb|400 px|Figure 2. Effect of Freundlich ''n'' on ''R''.]] | ||
The average rate that a contaminant migrates in groundwater is reduced because the sorbed portion of the contaminant does not migrate. The impact of sorption on the average contaminant transport velocity can be estimated using a Retardation Factor (''R''): | The average rate that a contaminant migrates in groundwater is reduced because the sorbed portion of the contaminant does not migrate. The impact of sorption on the average contaminant transport velocity can be estimated using a Retardation Factor (''R''): | ||
| + | [[File:Allen-King1w2 Eq3.png|center|620 px]] | ||
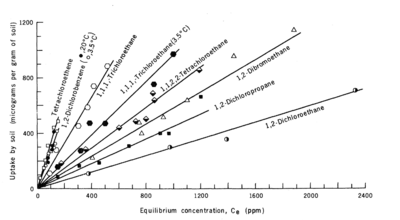
| − | + | [[File:Allen-King1w2 Fig3.png|thumb|400 px|Figure 3. Linear correlation between organic carbon content (''f<sub>oc</sub>'') and sorption distribution coefficient (''K<sub>d</sub>'') for four chlorinated aromatic contaminants<ref name="Schwarzenbach1981" />. ]] | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | [[File:Allen-King1w2 Fig3.png|thumb|400 px|Figure 3. Linear correlation between organic carbon content (''f<sub>oc</sub>'') and sorption distribution coefficient (<sub> | ||
When sorption of the contaminant is described by a linear isotherm and sorption processes are fast relative to water flow, ''R'' can be estimated as: | When sorption of the contaminant is described by a linear isotherm and sorption processes are fast relative to water flow, ''R'' can be estimated as: | ||
| − | [[File:Allen-King1w2 Eq4.png|center| | + | [[File:Allen-King1w2 Eq4.png|center|440 px]] |
| − | where | + | where ''ρ<sub>b</sub>'' is the bulk density of the solid phase and ''θ<sub>w</sub>'' is the water filled porosity. When sorption is non-linear and is described with the Freundlich isotherm equation, ''R'' can be estimated as: |
[[File:Allen-King1w2 Eq5.png|center|400px]] | [[File:Allen-King1w2 Eq5.png|center|400px]] | ||
| + | |||
| + | Figure 2 illustrates the effect of ''C<sub>w</sub>'' on ''R'' when 1/''n'' is less than 1 and ''ρ<sub>b</sub>'' = 1.5 g/cm<sup>3</sup>'', θ<sub>w</sub> = ''0.3 mL/cm<sup>3</sup>and ''K<sub>F</sub> = 2.'' When 1''/n'' is less than 1, sorption has limited effect in slowing the migration of a high concentration plume (''C<sub>w</sub>'' > 10 mg/L). However, as the aquifer is remediated and contaminant concentrations drop below 0.1 mg/L, non-linear sorption has a much more pronounced impact, slowing aquifer cleanup. | ||
==''K<sub>d</sub>'' Estimation Methods== | ==''K<sub>d</sub>'' Estimation Methods== | ||
| − | Multiple studies<ref name= "Schwarzenbach1981">Schwarzenbach, R.P. and Westall, J., 1981. Transport of nonpolar organic compounds from surface water to groundwater. Laboratory sorption studies. Environmental Science & Technology, 15(11), pp.1360-1367. [http://dx.doi.org/10.1021/es00093a009 | + | Multiple studies<ref name="Schwarzenbach1981">Schwarzenbach, R.P. and Westall, J., 1981. Transport of nonpolar organic compounds from surface water to groundwater. Laboratory sorption studies. Environmental Science & Technology, 15(11), pp.1360-1367. [http://dx.doi.org/10.1021/es00093a009 doi: 10.1021/es00093a009]</ref><ref>Karickhoff, S.W., 1981. Semi-empirical estimation of sorption of hydrophobic pollutants on natural sediments and soils. Chemosphere, 10(8), pp.833-846. [http://dx.doi.org/10.1016/0045-6535(81)90083-7 doi:10.1016/0045-6535(81)90083-7]</ref><ref>Chiou, C.T., Peters, L.J. and Freed, V.H., 1979. A physical concept of soil-water equilibria for nonionic organic compounds. Science, 206(4420), pp.831-832. [https://doi.org/10.1126/science.206.4420.831 doi: 10.1126/science.206.4420.831]</ref> showed that sorption of nonpolar organic contaminants is typically proportional to the organic carbon content of the solid phase (Figure 3). When this occurs, an organic carbon normalized partitioning coefficient, ''K<sub>oc</sub>'', is used: |
[[File:Allen-King1w2 Eq6.png|center|300 px]] | [[File:Allen-King1w2 Eq6.png|center|300 px]] | ||
| Line 66: | Line 68: | ||
where ''f<sub>oc</sub>'' is the fractional organic carbon content of the soil, sediment or rock. This approach empirically separates variations in ''K<sub>d</sub>'' according to variations in the organic matter concentration, an important property of the sorbent, and the ''K<sub>oc</sub>'', which is intended to capture differences in the chemical properties of the contaminant. | where ''f<sub>oc</sub>'' is the fractional organic carbon content of the soil, sediment or rock. This approach empirically separates variations in ''K<sub>d</sub>'' according to variations in the organic matter concentration, an important property of the sorbent, and the ''K<sub>oc</sub>'', which is intended to capture differences in the chemical properties of the contaminant. | ||
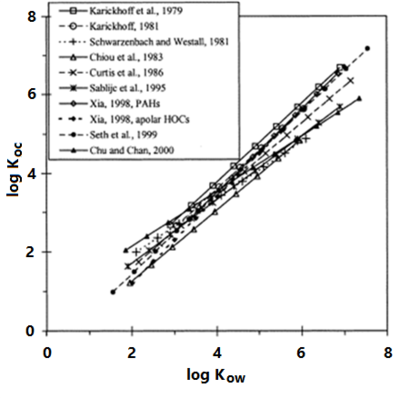
| − | Experimental investigations (summarized in Allen-King et al., 2002<ref name= "Allenking2002">Allen-King, R.M., Grathwohl, P. and Ball, W.P., 2002. New modeling paradigms for the sorption of hydrophobic organic chemicals to heterogeneous carbonaceous matter in soils, sediments, and rocks. Advances in Water Resources, 25(8), pp.985-1016. [http://dx.doi.org/10.1016/S0309-1708(02)00045-3 doi: 10.1016/S0309-1708(02)00045-3]</ref>) with a variety of sorbents and contaminants has shown that ''K<sub>oc</sub>'' values are correlated with contaminant hydrophobicity. This allows ''K<sub>oc</sub>'' to be estimated using empirical correlations with the contaminant aqueous solubility or [ | + | Experimental investigations (summarized in Allen-King et al., 2002<ref name="Allenking2002">Allen-King, R.M., Grathwohl, P. and Ball, W.P., 2002. New modeling paradigms for the sorption of hydrophobic organic chemicals to heterogeneous carbonaceous matter in soils, sediments, and rocks. Advances in Water Resources, 25(8), pp.985-1016. [http://dx.doi.org/10.1016/S0309-1708(02)00045-3 doi: 10.1016/S0309-1708(02)00045-3]</ref>) with a variety of sorbents and contaminants has shown that ''K<sub>oc</sub>'' values are correlated with contaminant hydrophobicity. This allows ''K<sub>oc</sub>'' to be estimated using empirical correlations with the contaminant aqueous solubility or [[wikipedia: Partition coefficient | octanol-water partition coefficient]] (''K<sub>ow</sub>'') (Figure 4). Using this approach, ''K<sub>d</sub>'' can be estimated using site specific measurements of ''f<sub>oc</sub>'' and literature values of contaminant properties. |
| − | [[File:Allen-King1w2 Fig4.png|thumb| | + | |
| + | [[File:Allen-King1w2 Fig4.png|thumb|left|400 px| Figure 4. Empirical correlations between log ''K<sub>oc</sub>'' and log ''K<sub>ow</sub>''. Variations arise because the studies used different contaminant compounds and different field samples. Figure from Allen-King et al., 2002<ref name="Allenking2002" />, which also lists the correlation equations.]] | ||
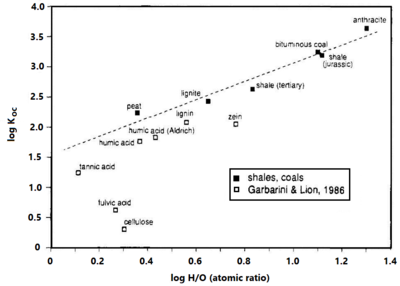
| − | [[File:Allen-King1w2 Fig5.png|thumb|right|400 px|Figure 5. The trichloroethene ''K<sub>oc</sub>'' versus the Hydrogen/Oxygen ratio of carbonaceous material. The sorption coefficient (per gram carbon) is 1-2 orders of magnitude greater for more condensed carbonaceous matter, as indicated by greater H/O ratios, compared to humic acid and peat. Calculations are from linear isotherms at a low aqueous concentration<ref name= "Grathwohl1990"/>.]] | + | [[File:Allen-King1w2 Fig5.png|thumb|right|400 px|Figure 5. The trichloroethene ''K<sub>oc</sub>'' versus the Hydrogen/Oxygen ratio of carbonaceous material. The sorption coefficient (per gram carbon) is 1-2 orders of magnitude greater for more condensed carbonaceous matter, as indicated by greater H/O ratios, compared to humic acid and peat. Calculations are from linear isotherms at a low aqueous concentration<ref name="Grathwohl1990" />.]] |
==Effect of Carbon Type== | ==Effect of Carbon Type== | ||
| − | Research reviewed by Allen-King et al. (2002)<ref name= "Allenking2002"/> Cornelissen et al. (2004)<ref name= "Cornelissen2004">Cornelissen, G., Kukulska, Z., Kalaitzidis, S., Christanis, K. and Gustafsson, Ö., 2004. Relations between environmental black carbon sorption and geochemical sorbent characteristics. Environmental Science & Technology, 38(13), pp.3632-3640. [http://dx.doi.org/10.1021/es0498742 doi: 10.1021/es0498742]</ref>, and Huang et al. (2003)<ref name= "Huang2003">Huang, W., Peng, P.A., Yu, Z. and Fu, J., 2003. Effects of organic matter heterogeneity on sorption and desorption of organic contaminants by soils and sediments. Applied Geochemistry, 18(7), pp.955-972. [http://dx.doi.org/10.1016/S0883-2927(02)00205-6 doi: 10.1016/S0883-2927(02)00205-6]</ref> indicates that some types of organic matter are much stronger sorbents than others. Experimental results show that thermally altered carbonaceous materials (TACMs), such as coals, kerogen and black carbons, will sorb greater concentrations of nonpolar organic contaminants than unaltered organic materials per gram of carbon ( | + | Research reviewed by Allen-King et al. (2002)<ref name="Allenking2002" /> Cornelissen et al. (2004)<ref name="Cornelissen2004">Cornelissen, G., Kukulska, Z., Kalaitzidis, S., Christanis, K. and Gustafsson, Ö., 2004. Relations between environmental black carbon sorption and geochemical sorbent characteristics. Environmental Science & Technology, 38(13), pp.3632-3640. [http://dx.doi.org/10.1021/es0498742 doi: 10.1021/es0498742]</ref>, and Huang et al. (2003)<ref name="Huang2003">Huang, W., Peng, P.A., Yu, Z. and Fu, J., 2003. Effects of organic matter heterogeneity on sorption and desorption of organic contaminants by soils and sediments. Applied Geochemistry, 18(7), pp.955-972. [http://dx.doi.org/10.1016/S0883-2927(02)00205-6 doi: 10.1016/S0883-2927(02)00205-6]</ref> indicates that some types of organic matter are much stronger sorbents than others. Experimental results show that thermally altered carbonaceous materials (TACMs), such as coals, kerogen and black carbons, will sorb greater concentrations of nonpolar organic contaminants than unaltered organic materials per gram of carbon (Figure 5), especially at low concentrations relative to solubility. TACMs are more condensed than their unaltered precursors, and have a lower oxygen content, and higher aromatic content than unaltered materials. Sorbed concentrations to TACM can be 10 to 100 times greater at equilibrium than the amount sorbed to less condensed forms of organic matter on a per-gram-carbon basis. |
| − | Sorption to TACMs is dominated by adsorptive behavior with strong sorption at low aqueous concentrations, with progressively weaker sorption at higher concentrations<ref name= "Cornelissen2004"/><ref name= "Allenking2002"/><ref name= "Huang2003"/>. This effect is commonly represented with a Freundlich isotherm with the Freundlich exponent <1. | + | Sorption to TACMs is dominated by adsorptive behavior with strong sorption at low aqueous concentrations, with progressively weaker sorption at higher concentrations<ref name="Cornelissen2004" /><ref name="Allenking2002" /><ref name="Huang2003" />. This effect is commonly represented with a Freundlich isotherm with the Freundlich exponent <1. |
| − | TACMs are typically associated with shale rocks or coal containing kerogen that has been buried and lithified, or with soils containing char or soot from incomplete combustion. This material may be present in consolidated rock or in sediments that contain these materials<ref>Kleineidam, S., Rügner, H., Ligouis, B. and Grathwohl, P., 1999. Organic matter facies and equilibrium sorption of phenanthrene. Environmental Science & Technology, 33(10), pp.1637-1644. [http://dx.doi.org/10.1021/es9806635 doi: 10.1021/es9806635]</ref><ref>Binger, C.A., Martin, J.P., Allen-King, R.M. and Fowler, M., 1999. Variability of chlorinated-solvent sorption associated with oxidative weathering of kerogen. Journal of Contaminant Hydrology, 40(2), pp.137-158. [http://dx.doi.org/10.1016/S0169-7722(99)00047-9 doi: 10.1016/S0169-7722(99)00047-9]</ref>. However, weathering of these materials appears to diminish adsorption<ref name= "Grathwohl1990"> Grathwohl, P., 1990. Influence of organic matter from soils and sediments from various origins on the sorption of some chlorinated aliphatic hydrocarbons: implications on Koc correlations. Environmental Science & Technology, 24(11), pp.1687-1693. [http://dx.doi.org/10.1021/es00081a010 doi: 10.1021/es00081a010]</ref>. | + | TACMs are typically associated with shale rocks or coal containing kerogen that has been buried and lithified, or with soils containing char or soot from incomplete combustion. This material may be present in consolidated rock or in sediments that contain these materials<ref>Kleineidam, S., Rügner, H., Ligouis, B. and Grathwohl, P., 1999. Organic matter facies and equilibrium sorption of phenanthrene. Environmental Science & Technology, 33(10), pp.1637-1644. [http://dx.doi.org/10.1021/es9806635 doi: 10.1021/es9806635]</ref><ref>Binger, C.A., Martin, J.P., Allen-King, R.M. and Fowler, M., 1999. Variability of chlorinated-solvent sorption associated with oxidative weathering of kerogen. Journal of Contaminant Hydrology, 40(2), pp.137-158. [http://dx.doi.org/10.1016/S0169-7722(99)00047-9 doi: 10.1016/S0169-7722(99)00047-9]</ref>. However, weathering of these materials appears to diminish adsorption<ref name="Grathwohl1990"> Grathwohl, P., 1990. Influence of organic matter from soils and sediments from various origins on the sorption of some chlorinated aliphatic hydrocarbons: implications on Koc correlations. Environmental Science & Technology, 24(11), pp.1687-1693. [http://dx.doi.org/10.1021/es00081a010 doi: 10.1021/es00081a010]</ref>. |
==How to Estimate Sorption== | ==How to Estimate Sorption== | ||
| − | For high contaminant concentrations or when amorphous organic matter dominates sorption, measurements of soil ''f<sub>oc</sub>'' and empirical correlations with contaminant properties appear to provide ''K<sub>d</sub>'' estimates within about a factor of 3 of values measured in laboratory isotherms<ref name= "Allenking2002"/>. However, when TACMs are present and contaminant concentrations are low, the empirical correlations may substantially underestimate sorption. Additional research is needed to determine when simple empirical correlations may be used to estimate ''K<sub>d</sub>'' and when more rigorous experimental investigations are required. | + | For high contaminant concentrations or when amorphous organic matter dominates sorption, measurements of soil ''f<sub>oc</sub>'' and empirical correlations with contaminant properties appear to provide ''K<sub>d</sub>'' estimates within about a factor of 3 of values measured in laboratory isotherms<ref name="Allenking2002" />. However, when TACMs are present and contaminant concentrations are low, the empirical correlations may substantially underestimate sorption. Additional research is needed to determine when simple empirical correlations may be used to estimate ''K<sub>d</sub>'' and when more rigorous experimental investigations are required. |
==Summary== | ==Summary== | ||
| − | Nonionic and nonpolar organic contaminants including chlorinated solvents and petroleum hydrocarbons can become associated with solid phase organic matter in the soil, sediment, and rock. When amorphous organic matter is the primary sorbent, sorption is linear and distribution coefficients can be estimated from empirical correlations with contaminant properties (aqueous solubility or octanol-water partition coefficient) and site-specific measurements of solid phase organic carbon. However, when thermally altered carbonaceous materials (TACMs) are present, sorption may be non-linear and the empirical correlations may substantially underestimate sorption at low contaminant concentrations. | + | Nonionic and nonpolar organic contaminants including chlorinated solvents and petroleum hydrocarbons can become associated with solid phase organic matter in the soil, sediment, and rock. When amorphous organic matter is the primary sorbent, sorption is linear and distribution coefficients can be estimated from empirical correlations with contaminant properties (aqueous solubility or octanol-water partition coefficient) and site-specific measurements of solid phase organic carbon. However, when thermally altered carbonaceous materials (TACMs) are present, sorption may be non-linear and the empirical correlations may substantially underestimate sorption at low contaminant concentrations. |
| + | <br clear="right"> | ||
==References== | ==References== | ||
Revision as of 16:07, 16 May 2019
Nonionic and nonpolar organic contaminants including chlorinated solvents and petroleum hydrocarbons can become associated with solid phase organic matter in the soil, sediment, and rock, slowing contaminant migration in groundwater and reducing the efficacy of many remedial technologies. When amorphous organic matter is the primary sorbent, sorption is linear and distribution coefficients can be estimated from empirical correlations with contaminant properties (aqueous solubility or octanol-water partition coefficient) and site specific measurements of solid phase organic carbon. However, when thermally altered carbonaceous materials (TACMs) are present, sorption may be non-linear and the empirical correlations may substantially underestimate sorption at low contaminant concentrations.
Related Article(s):
CONTRIBUTOR(S): Richelle Allen-King
Key Resource(s):
Introduction
Sorption is a non-mechanistic term that describes uptake of a contaminant by a solid phase sorbent (soil, sediment, rock or an engineered material such as activated carbon). This article focuses on sorption of nonionic and nonpolar organic contaminants and their associations with subsurface materials. For these contaminants, sorption is almost exclusively associated with organic matter in the solids and is a reversible process. Sorption of ionic and polar organic contaminants is not described in this article.

In this article, the term ‘sorption’ includes all processes that cause the dissolved organic compound (sorbate) to become associated with the solid phase (sorbent). For nonpolar organic compounds, sorption occurs by either adsorption or absorption. Absorption is a volume-based process whereby the sorbate diffuses into the organic matter, analogous to the way that organic contaminants partition between immiscible oil and aqueous phases. In contrast, adsorption describes the interaction between the contaminant and the sorbent surfaces.
Measuring Sorption
Sorption can be measured in the laboratory by placing a known mass of the contaminant, water, and uncontaminated sediment in a batch reactor (Figure 1a). The system is mixed for a specific duration (contact time) and then the aqueous and sorbed contaminant concentrations are determined. This process is repeated, adding a range of different initial contaminant masses to achieve a range of aqueous contaminant concentrations. When the experiment is run at constant temperature until the concentrations in the solid and liquid phases approach equilibrium, the resulting data plot is termed an isotherm. Figure 1b shows linear sorption isotherms for eight compounds measured in one soil.
The time required to reach equilibrium can vary depending on the contaminant and sorbent. In studies with aquifer material from Canadian Forces Base Borden, tetrachloroethene (PCE) sorption reached equilibrium within 2 - 4 weeks, but tetrachlorobenzene did not reach equilibrium after 100 days. Powdered (pulverized) samples equilibrated more quickly indicating mass transfer within the grains was limiting[3].
In some cases, the experimental data show an approximately linear relationship between aqueous and sorbed concentration, and the experimental results can be summarized in the form of a partition or distribution coefficient, Kd:
where CS is the solid (sorbed) concentration and Cw is the aqueous (dissolved) concentration. By convention, Cs has units of µg/g and Cs has units of µg/mL, so Kd has units of mL/g.
When the relationship between Cs and Cw is non-linear, the experimental data are commonly represented by the Freundlich isotherm equation:
where KF and n are empirically estimated coefficients specific to the sorbate, sorbent and equation used.
In most cases, absorption processes follow a linear isotherm. However, adsorption processes often follow a nonlinear relationship between the sorbed and dissolved contaminant concentrations. When both absorption and adsorption occur, a non-linear relationship is used to represent the sorption results.
Impact on Groundwater Transport
Sorption reduces the amount of contaminant in the aqueous phase, slowing contaminant migration. Sorbed contaminants are also less available for biological or chemical degradation reactions, which will reduce the effectiveness of many remedial technologies.
The average rate that a contaminant migrates in groundwater is reduced because the sorbed portion of the contaminant does not migrate. The impact of sorption on the average contaminant transport velocity can be estimated using a Retardation Factor (R):
When sorption of the contaminant is described by a linear isotherm and sorption processes are fast relative to water flow, R can be estimated as:
where ρb is the bulk density of the solid phase and θw is the water filled porosity. When sorption is non-linear and is described with the Freundlich isotherm equation, R can be estimated as:
Figure 2 illustrates the effect of Cw on R when 1/n is less than 1 and ρb = 1.5 g/cm3, θw = 0.3 mL/cm3and KF = 2. When 1/n is less than 1, sorption has limited effect in slowing the migration of a high concentration plume (Cw > 10 mg/L). However, as the aquifer is remediated and contaminant concentrations drop below 0.1 mg/L, non-linear sorption has a much more pronounced impact, slowing aquifer cleanup.
Kd Estimation Methods
Multiple studies[4][5][6] showed that sorption of nonpolar organic contaminants is typically proportional to the organic carbon content of the solid phase (Figure 3). When this occurs, an organic carbon normalized partitioning coefficient, Koc, is used:
where foc is the fractional organic carbon content of the soil, sediment or rock. This approach empirically separates variations in Kd according to variations in the organic matter concentration, an important property of the sorbent, and the Koc, which is intended to capture differences in the chemical properties of the contaminant.
Experimental investigations (summarized in Allen-King et al., 2002[7]) with a variety of sorbents and contaminants has shown that Koc values are correlated with contaminant hydrophobicity. This allows Koc to be estimated using empirical correlations with the contaminant aqueous solubility or octanol-water partition coefficient (Kow) (Figure 4). Using this approach, Kd can be estimated using site specific measurements of foc and literature values of contaminant properties.
Effect of Carbon Type
Research reviewed by Allen-King et al. (2002)[7] Cornelissen et al. (2004)[9], and Huang et al. (2003)[10] indicates that some types of organic matter are much stronger sorbents than others. Experimental results show that thermally altered carbonaceous materials (TACMs), such as coals, kerogen and black carbons, will sorb greater concentrations of nonpolar organic contaminants than unaltered organic materials per gram of carbon (Figure 5), especially at low concentrations relative to solubility. TACMs are more condensed than their unaltered precursors, and have a lower oxygen content, and higher aromatic content than unaltered materials. Sorbed concentrations to TACM can be 10 to 100 times greater at equilibrium than the amount sorbed to less condensed forms of organic matter on a per-gram-carbon basis.
Sorption to TACMs is dominated by adsorptive behavior with strong sorption at low aqueous concentrations, with progressively weaker sorption at higher concentrations[9][7][10]. This effect is commonly represented with a Freundlich isotherm with the Freundlich exponent <1.
TACMs are typically associated with shale rocks or coal containing kerogen that has been buried and lithified, or with soils containing char or soot from incomplete combustion. This material may be present in consolidated rock or in sediments that contain these materials[11][12]. However, weathering of these materials appears to diminish adsorption[8].
How to Estimate Sorption
For high contaminant concentrations or when amorphous organic matter dominates sorption, measurements of soil foc and empirical correlations with contaminant properties appear to provide Kd estimates within about a factor of 3 of values measured in laboratory isotherms[7]. However, when TACMs are present and contaminant concentrations are low, the empirical correlations may substantially underestimate sorption. Additional research is needed to determine when simple empirical correlations may be used to estimate Kd and when more rigorous experimental investigations are required.
Summary
Nonionic and nonpolar organic contaminants including chlorinated solvents and petroleum hydrocarbons can become associated with solid phase organic matter in the soil, sediment, and rock. When amorphous organic matter is the primary sorbent, sorption is linear and distribution coefficients can be estimated from empirical correlations with contaminant properties (aqueous solubility or octanol-water partition coefficient) and site-specific measurements of solid phase organic carbon. However, when thermally altered carbonaceous materials (TACMs) are present, sorption may be non-linear and the empirical correlations may substantially underestimate sorption at low contaminant concentrations.
References
- ^ Piwoni, M.D. and Keeley, J.W., 1990. Basic concepts of contaminant sorption at hazardous waste sites. Ground-Water Issue; EPA-540/4-90/053. Report.pdf
- ^ 2.0 2.1 Chiou, C.T., Porter, P.E. and Schmedding, D.W., 1983. Partition equilibriums of nonionic organic compounds between soil organic matter and water. Environmental Science & Technology, 17(4), pp.227-231. doi: 10.1021/es00110a009
- ^ Ball, W.P. and Roberts, P.V., 1991. Long-term sorption of halogenated organic chemicals by aquifer material. 2. Intraparticle diffusion. Environmental Science & Technology, 25(7), pp.1237-1249. doi:10.1021/es00019a003
- ^ 4.0 4.1 Schwarzenbach, R.P. and Westall, J., 1981. Transport of nonpolar organic compounds from surface water to groundwater. Laboratory sorption studies. Environmental Science & Technology, 15(11), pp.1360-1367. doi: 10.1021/es00093a009
- ^ Karickhoff, S.W., 1981. Semi-empirical estimation of sorption of hydrophobic pollutants on natural sediments and soils. Chemosphere, 10(8), pp.833-846. doi:10.1016/0045-6535(81)90083-7
- ^ Chiou, C.T., Peters, L.J. and Freed, V.H., 1979. A physical concept of soil-water equilibria for nonionic organic compounds. Science, 206(4420), pp.831-832. doi: 10.1126/science.206.4420.831
- ^ 7.0 7.1 7.2 7.3 7.4 Allen-King, R.M., Grathwohl, P. and Ball, W.P., 2002. New modeling paradigms for the sorption of hydrophobic organic chemicals to heterogeneous carbonaceous matter in soils, sediments, and rocks. Advances in Water Resources, 25(8), pp.985-1016. doi: 10.1016/S0309-1708(02)00045-3
- ^ 8.0 8.1 Grathwohl, P., 1990. Influence of organic matter from soils and sediments from various origins on the sorption of some chlorinated aliphatic hydrocarbons: implications on Koc correlations. Environmental Science & Technology, 24(11), pp.1687-1693. doi: 10.1021/es00081a010
- ^ 9.0 9.1 Cornelissen, G., Kukulska, Z., Kalaitzidis, S., Christanis, K. and Gustafsson, Ö., 2004. Relations between environmental black carbon sorption and geochemical sorbent characteristics. Environmental Science & Technology, 38(13), pp.3632-3640. doi: 10.1021/es0498742
- ^ 10.0 10.1 Huang, W., Peng, P.A., Yu, Z. and Fu, J., 2003. Effects of organic matter heterogeneity on sorption and desorption of organic contaminants by soils and sediments. Applied Geochemistry, 18(7), pp.955-972. doi: 10.1016/S0883-2927(02)00205-6
- ^ Kleineidam, S., Rügner, H., Ligouis, B. and Grathwohl, P., 1999. Organic matter facies and equilibrium sorption of phenanthrene. Environmental Science & Technology, 33(10), pp.1637-1644. doi: 10.1021/es9806635
- ^ Binger, C.A., Martin, J.P., Allen-King, R.M. and Fowler, M., 1999. Variability of chlorinated-solvent sorption associated with oxidative weathering of kerogen. Journal of Contaminant Hydrology, 40(2), pp.137-158. doi: 10.1016/S0169-7722(99)00047-9